Hepatitis B infection model
Model description - HBV model
Nova’s hepatitis B disease (HBV) model accounts for the virus pathophysiology, including viral replication within hepatocytes and antiviral immune response in chronically infected patients. Nova's chronic hepatitis B model for exploring and predicting efficacy for novel HBV treatments.
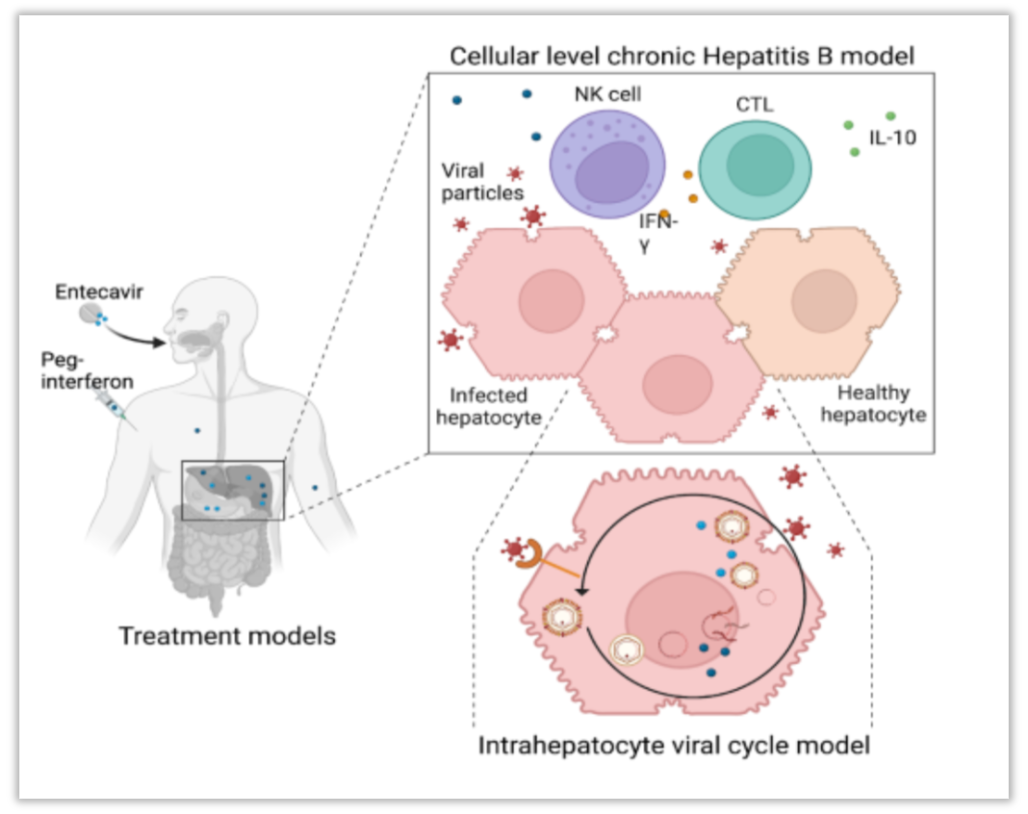
Structure of the HBV model. The patho-physiology model includes an intra-hepatocyte viral cycle submodel coupled with a cellular level chronic hepatitis B submodel for the dynamics of infected and healthy hepatocytes as well as immune cells and cytokines. Entecavir and peg-interferon alpha treatments models are coupled to the patho-physiology model to allow simulating patient’s response to treatments.
💊 Drugs that can be tested
-
Nucleos(t)ide analogs (NUC) (eg.
-
entecavir, tenofovir)
-
Direct antivirals (including small interfering RNA, entry inhibitors, capsid assembly modulators)
-
Indirect antivirals (including immunomodulators such as peg-interferon alfa, recombinant vaccines, FXR agonist)
👥 Model Populations of interest
- Adult chronic HBV population
- Treatment naive or NUC-controlled
- HBe antigen positive or negative
- Single of co-infection with HDV
- (Hepatitis Delta)
- Various HBV genotypes
📍Possible clinical endpoints
-
Viral markers (HBV DNA, HBsAg, HBeAg, HBV RNA) in serum
-
Immune markers (cytokines eg. IL-10, immune cells eg. CD8+ T cells, NKC)
-
Possible extension: liver cirrhosis, hepatic carcinoma, liver failure
Biological submodels
- HBV replication & excretion
- Bile acid metabolism
- Cholesterol synthesis and intake
- Impact of HBV on hepatocytes
- Immune system
- Drug model of your treatment
- Drug models of Standard of Care (entecavir, peg-IFN-alpha)
Early insights to optimize your trial design - HBV model
Drug Regimen
- What is the impact of the dose per administration of an investigational treatment on pharmacodynamics markers?
- What is the impact of treatment duration?
- What is the best follow-up duration to assess efficacy?
Combination of treatments
- What is the difference in efficacy of an investigational treatment dose when combining it with IFN treatment?
- What is the impact of adding ETV on top of the other 2 treatments?
- What is best between a full combination therapy approach and a sequential / alternating treatment
Patient selection
- What is the impact of various inclusion criteria (BeAg status, Hepatitis B viral genotype, age...)?
"Generating such insights upstream within a few months instead of many years reduces the risks, and make our drug development strategies eventually more successful."
Pietro SCALFARO
Chief Medical Officer, ENYO Pharma