Non-Small Cell Lung Cancer (NSCLC) Model
Model description - NSCLC
Nova's non small cell lung cancer (NSCLC) disease model of epidermal growth factor receptor (EGFR) mutant lung adenocarcinoma (LUAD) includes intracellular signalling pathways, different metastatic sites, tumor cell dynamics with the immune system and tumor evolution. It allows investigating new treatments against EGFR mutant LUAD.
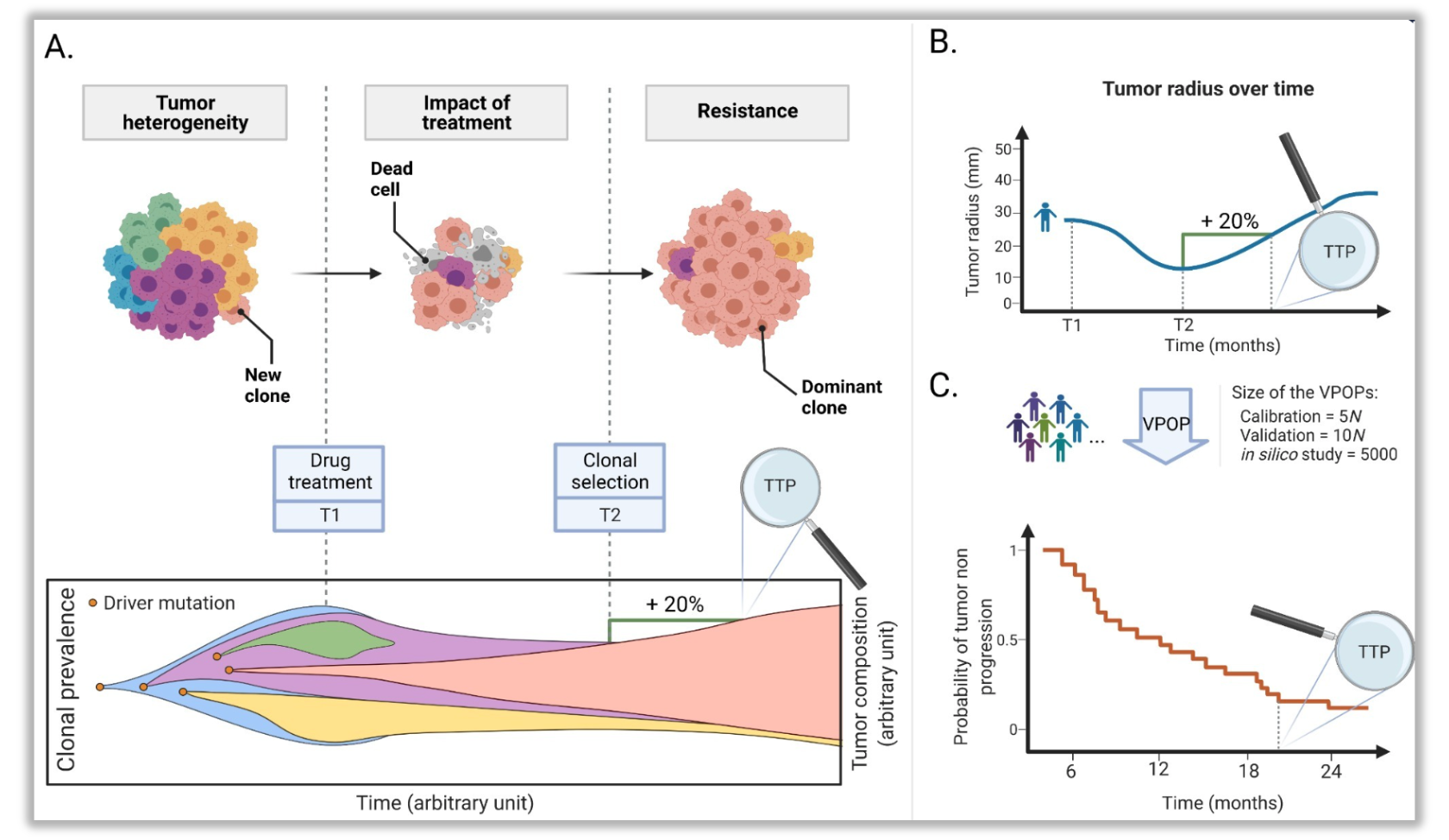
Quantification of tumor size evolution affected by clonal prevalence. A. Tumor growth and heterogeneity. B. Tumor radius evolution computed on each virtual patient and the time to progression is deduced from RECIST criteria. C. Tumor radius is followed for each patient of the virtual population and represented with a Kaplan-Meier visualization of theprobability of tumor non-progression .
Biological submodels
- EGFR signaling pathways
- Cell cycle and cell death
- Neoangiogenesis
- Immune system and its impact on tumor
- Tumor genotype and phenotype
- Clinical evaluation of tumor progression
- Treatment with standard of care (e.g. TKI, chemotherapy, immunotherapy)
- Your treatment(s) of interest
💊 Drugs that can be tested
-
Kinase inhibitors (TKI, cyclin-dependent, etc.)
-
Co-medication with standard platinum-based chemotherapies
-
Inhibitory antibodies
-
IO-therapies
-
Antibody–drug conjugates (ADCs)
-
Antiangiogenic therapies
-
Metabolic inhibitors
-
Telomerase inhibitors
-
Pro-apoptotic therapies
👥 Model Populations of interest
Adults
- EGFR mutant LUAD at stage Illb or IV
- Possible extension: stage ll and Illa LUAD, NSCLC
📍Possible clinical endpoints
- Time to progression (TTP), according to RECIST 1.1 criteria
- Objective response rate (ORR)
- Metastase appearance
- Tumor size (longest dimension or volume) for primary tumor and metastases
Early insights to optimize your trial design - NSCLC model
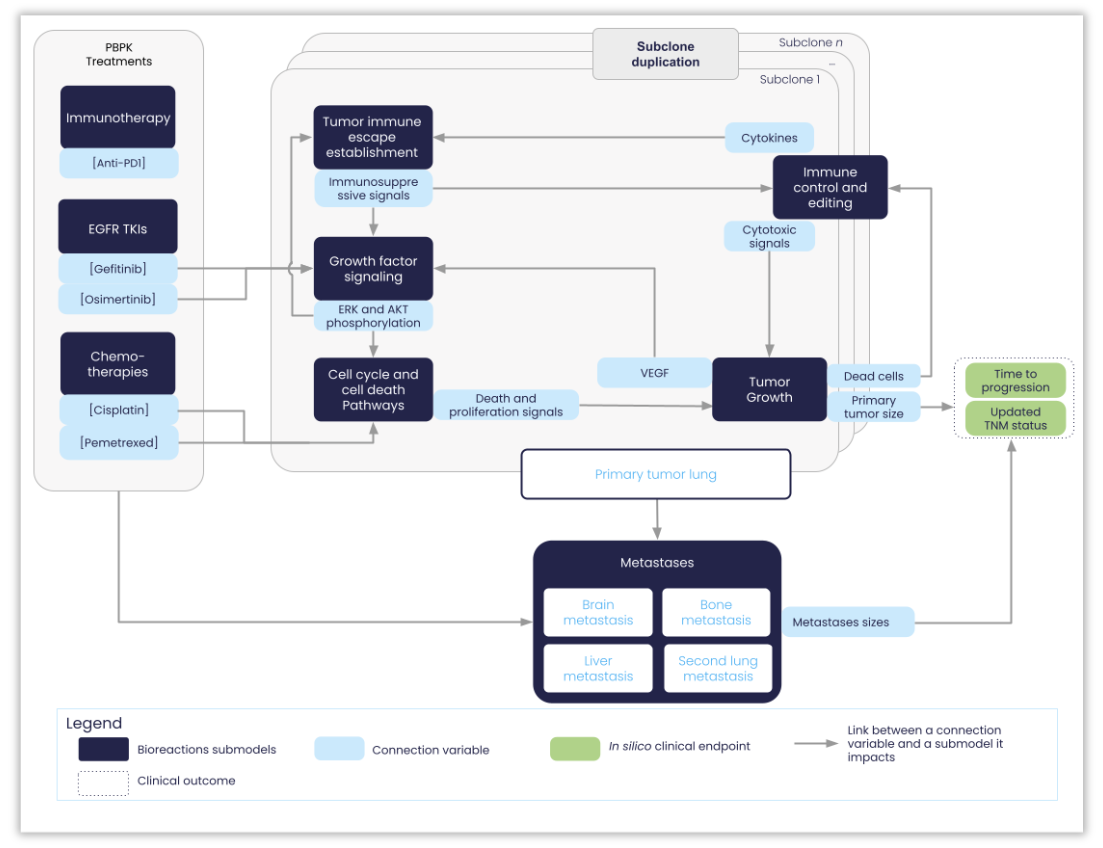
Structure of the NSCLC-V2 model: the different submodels are labeled and their connecting variables are represented in light blue. The two main model outputs are also represented (i) the biological one, corresponding to the radius of the primary & metastases tumors; (ii) the clinical one, corresponding to the time at which the disease progressed, as defined according to the RECIST (Response Evaluation Criteria In Solid Tumors) guidelines (version 1.1) .
"We’ve collaborated with Nova on the development of the NSCLC mechanistic model for over three years, and we’re thrilled to see the simulation results validated on one of our studies. This predictive model can now be employed to simulate hypotheses that we cannot test in real life, especially in populations with rare driver mutations like the exon20 insertion on EGFR."
Mathias Bergeron
Medical Director in Oncology Janssen
Drug Regimen
- What is the optimal posology (dose and frequency of administration) to maximize patient response?
- What is the best duration of treatment?
Cost-effective upgrades
EGFR wild type Addition of different driver mutations Squamous cell carcinoma ORR Early stage OS
Trial design
- Use of synthetic control arms to evaluate the efficacy of a treatment
- Prediction of long term endpoints
- Inclusion / Exclusion criteria
Combination of treatments
- What is the best combination or sequence of treatments to maximize target patient response?
- How does the investigational treatment (alone or in combination with standard of care compare with standard of care?)
Patient selection
What are the patient characteristics (e.g. age, gender, tumor initial size and genetic background) impacting response to treatment?
"Nova's generated proof-of-concept of its predictive disease models through our preliminary work over the past year and we look forward to continuing our ground-breaking work"
Véronique Schmitt
Market Access, New Products and Governmental Affairs Director, Janssen